 Molecular solids composed of molecules with permanent dipole moments (polar molecules) melt at still higher temperatures. 
Substances consisting of larger, nonpolar molecules have larger attractive forces and melt at higher temperatures. Small symmetrical molecules (nonpolar molecules), such as H 2, N 2, O 2, and F 2, have weak attractive forces and form molecular solids with very low melting points (below −200 ☌). The strengths of the attractive forces between the units present in different crystals vary widely, as indicated by the melting points of the crystals. 
Molecular solids, such as ice, sucrose (table sugar), and iodine, as shown in Figure 10.42, are composed of neutral molecules. Unlike typical covalent solids, graphite is very soft and electrically conductive. Graphite is an exceptional example, composed of planar sheets of covalent crystals that are held together in layers by noncovalent forces. These differences reflect differences in strengths of metallic bonding among the metals.įigure 10.41 A covalent crystal contains a three-dimensional network of covalent bonds, as illustrated by the structures of diamond, silicon dioxide, silicon carbide, and graphite. Several post-transition metals also have low melting points, whereas the transition metals melt at temperatures above 1000 ☌. Mercury is a liquid at room temperature, and the alkali metals melt below 200 ☌. The melting points of the metals vary widely. Because of their malleability (the ability to deform under pressure or hammering), they do not shatter and, therefore, make useful construction materials. All exhibit high thermal and electrical conductivity, metallic luster, and malleability. The atoms within such a metallic solid are held together by a unique force known as metallic bonding that gives rise to many useful and varied bulk properties. The structure of metallic crystals is often described as a uniform distribution of atomic nuclei within a “sea” of delocalized electrons. 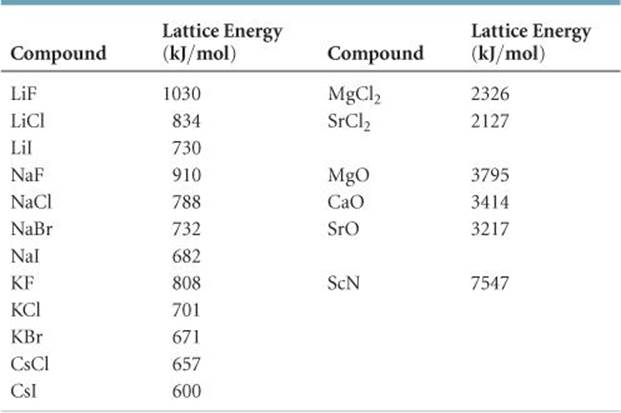
Metallic solids such as crystals of copper, aluminum, and iron are formed by metal atoms Figure 10.40. Many simple compounds formed by the reaction of a metallic element with a nonmetallic element are ionic.įigure 10.39 Sodium chloride is an ionic solid. Ionic solids do not conduct electricity however, they do conduct when molten or dissolved because their ions are free to move. Although they are hard, they also tend to be brittle, and they shatter rather than bend. This will be looked at in more detail in a later discussion of lattice energies. This is due to the very strong attractions between the ions-in ionic compounds, the attractions between full charges are (much) larger than those between the partial charges in polar molecular compounds. Many ionic crystals also have high melting points. Ionic solids, such as sodium chloride and nickel oxide, are composed of positive and negative ions that are held together by electrostatic attractions, which can be quite strong ( Figure 10.39). The following sections provide descriptions of the major types of crystalline solids: ionic, metallic, covalent network, and molecular. These forces are primarily responsible for the physical properties exhibited by the bulk solids. (b) Rapid cooling of molten SiO 2 yields an amorphous solid known as “fused silica”.Ĭrystalline solids are generally classified according to the nature of the forces that hold its particles together. 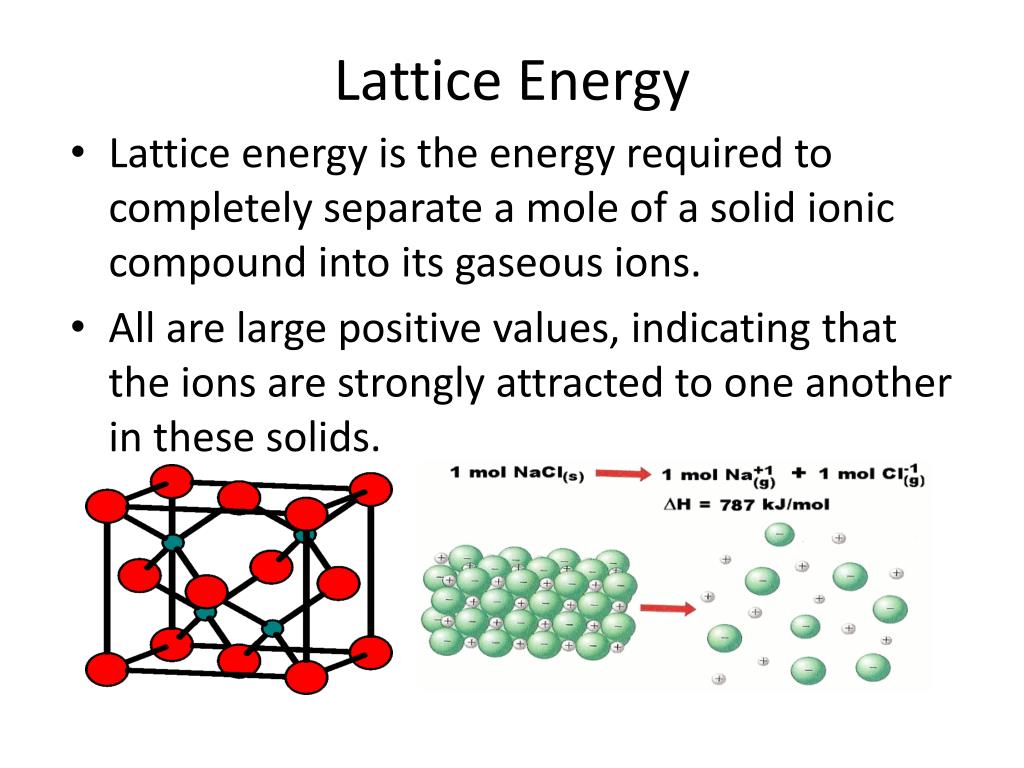
Figure 10.38 (a) Silicon dioxide, SiO 2, is abundant in nature as one of several crystalline forms of the mineral quartz. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
